Zeta-potential & Particle size Analyzer ELSZ-2000 series
|
|
In addition to existing zeta potential and particle size measurement, molecular weight analysis function is newly added. (Particle size :0.6nm-10um/Concentration range: 0.00001%-40%). Are you interested in alliance or distributorship for this product? |
- Product
- Principle
- Specification
- Measured Data
- Option
Product
- Higher sensitivity and shorter measure time with new APD.
- Melting point and phase transition point analysis enabled by auto temperature gradient function.
- Wide temperature range between 0 and 90℃.
- Wide range Molecular weight analysis.
- Supports concentrated sample measurement.
- High precision zeta potential analysis thanks to electro-osmosis measurement and plot analysis.
- Supports zeta potential measurement of salt condensed sample.
- Surface zeta potential of smaller area sample.
| Zeta Potential | No effective limitations | |||
| Mobility | -20×10 -4 ~ 20×10 -4 cm2/V・s | |||
| Particle Size | 0.6 nm ~ 10000 nm | |||
| Molecular Weight | 360 ~ 2000×104 | |||
●Measure Range
| Temperature | 0 ~ 90℃ | |||
| Concentration | Particle Size : 0.00001 % (0.1ppm) ~ 40 % *1 Zeta Potential : 0.001%~40% |
|||
*1(Latex112nm: 0.00001 ~ 10%、Taurocholic acid: ~ 40%)
Best suitable for basic and applied research of particle characterization in the field of surface chemical, inorganic, semiconductor, polymer, biotechnology, pharmaceutical and medical as well as surface research of film and flat state sample.
- New functional material
Fuel cell(Carbon nanotube, Fullerene, Function film, Catalyst, Nano-metal)
Bionanotechnology ( Nano capsule, Dendrimer, DDS), Nanobubble
- Ceramic and paints
Ceramic(Silica, Alumina, TiO2)
Surface modifier, dispersion and cohesion of inorganic sol
Dispersion and cohesion of carbon black and organic pigment
Slurry state sample
Color filter
Flotation adsorption research - Semiconductor
Alien objects research on silicon wafer
Interaction research of abrasive/additive on wafer surface
CMP slurry
- Polymer and Chemical
Emulsion dispersion and cohesion
Surface modifier of latex
Function research of polyelectrolyte
Process control of paper production and Pulp additive research
- Pharmaceutical and food
Emulsion dispersion and cohesion
Dispersion/Cohesion control of liposome and vesicle
Functionality of surfactant(Micelle)
Principle
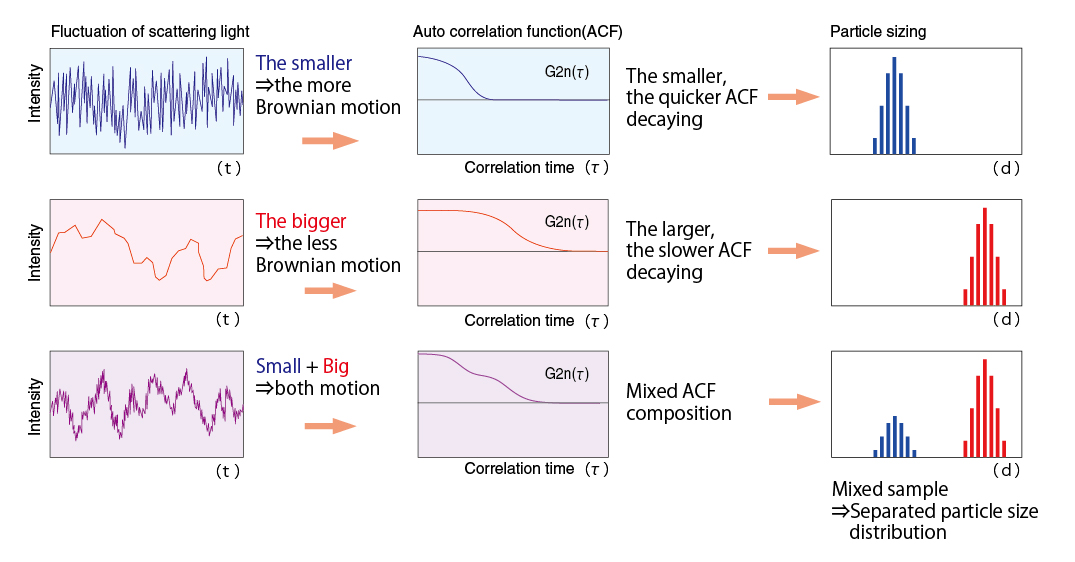
Particulates dispersed in a solution are normally subject to Brownian motion. The motion is slower with larger particles and faster with smaller particles. When laser light illuminates particles under the influence of Brownian motion, scattered light from the particles shows fluctuation corresponding to individual particles. The fluctuation is observed according to the pinhole type photon detection method so that particle size and particle size distributions are calculated.
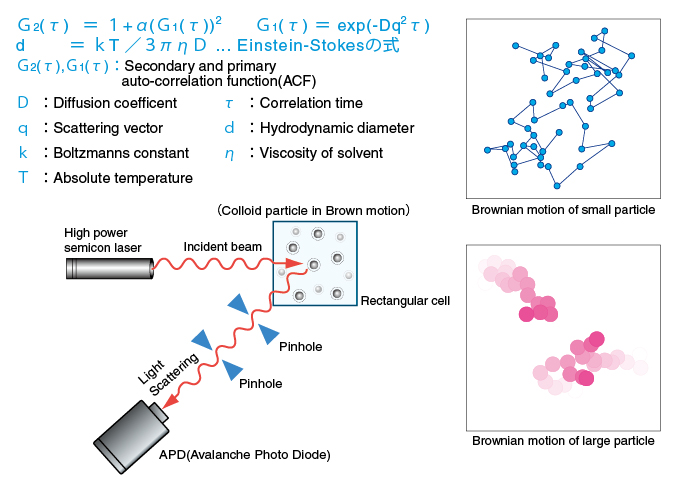
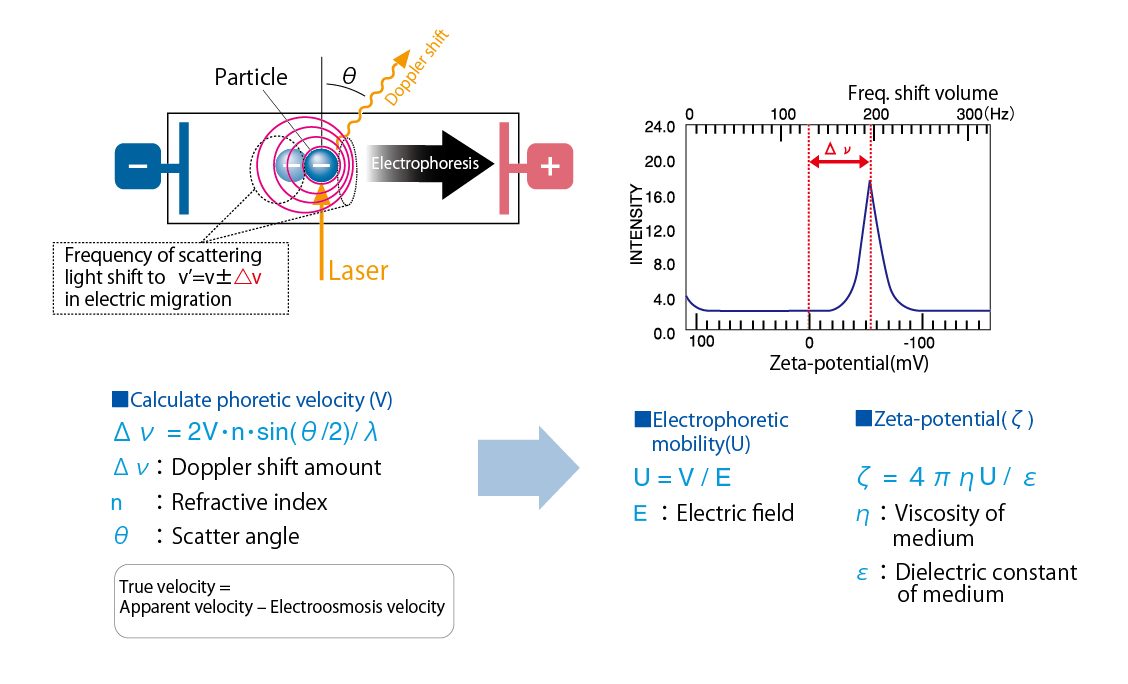
In most cases, colloidal particles possess a positive or negative electrostatic charge. As electrical fields are applied to the particle dispersion, the particles migrate in oppositely charged directions. As particles are irradiated in migration, scattering light causes Doppler shift depending on electrophoretic mobility.This method is called Laser Doppler Method.
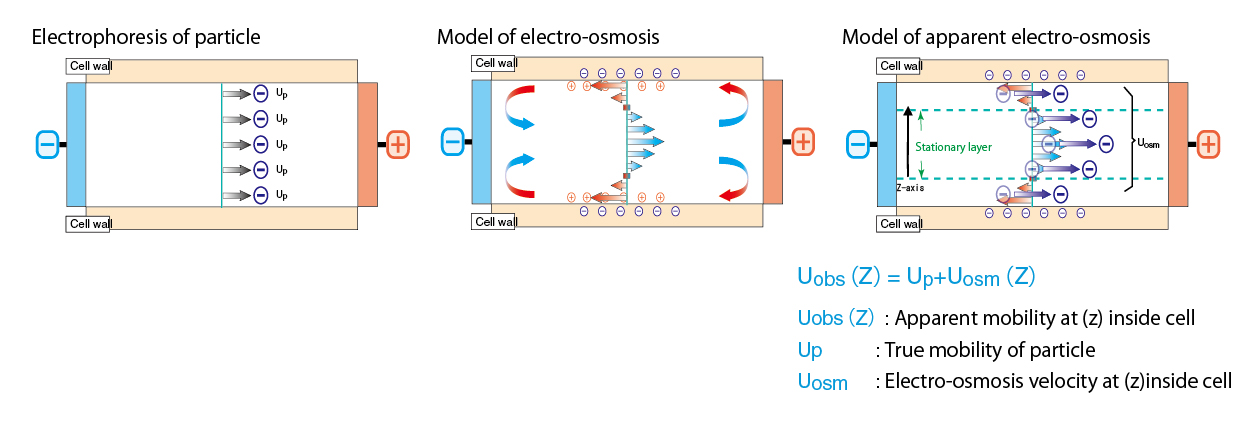
Electro-osmosis expresses the liquid flow occurred inside cell upon zeta potential measurement. If cell wall is electrically charged, counter ion in medium migrates to cell wall. This is the phenomenon that counter ion migrates to one electrode in the cell wall and to other electrode in the vicinity of cell center. By measuring apparent electrophoresis mobility and analyze electro osmosis, it become possible to obtain correct stationary layer taking stained cell into consideration and to calculate correct zeta potential. (Mori-Okamoto equation)
<Mori-Okamoto equation >
Uobs(z)=AU0(z/b)2+⊿U0(z/b)+(1-A)U0+Up
z:Distance from cell center
Uobs(z):Apparent mobility at (z) inside cell
A=1/[(2/3)-(0.420166/k)]
k=2a and 2b are respectively the horizontal and vertical length of square phase of cell. Here a>b.
Up:True mobility of particle
U0:Average mobility at upper and bottom walls
⊿U0:Gap of mobility at upper and bottom walls
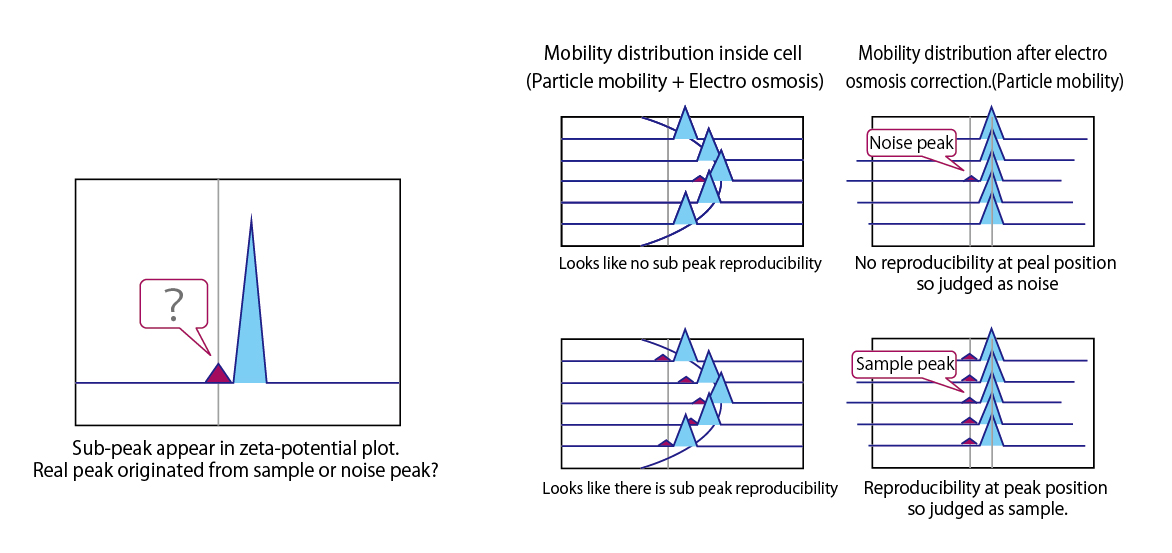
Electro-osmosis for multi component analysis
Apparent electro-osmosis measure at multiple points inside cell enables repeatability check of zeta potential and noise or peak determination.
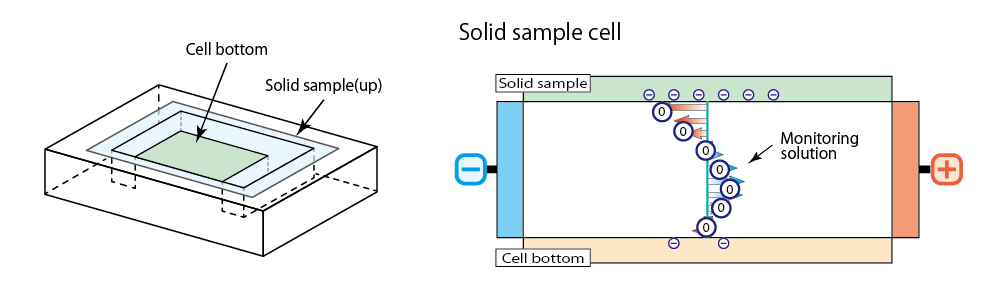
Solid sample cell is configured with box-like quartz cell with flat surface sample attached on it. Measure apparent electrophoresis mobility of monitoring particle at several positions in vertical inside cell and analyze mobility of electro osmosis on solid surface using electro osmosis profile obtained to calculate zeta potential.
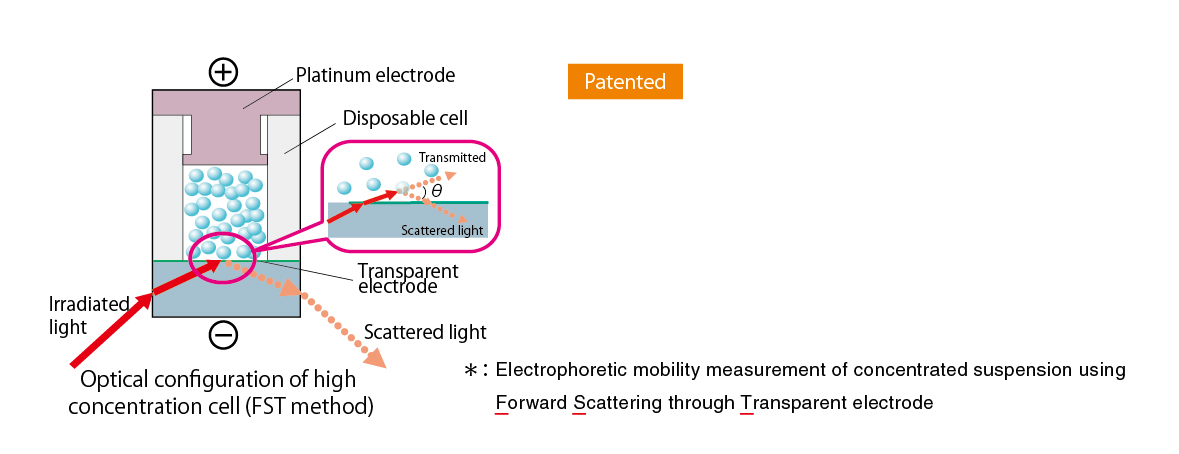
Principle of zeta-potential measurement using high concentration cell
It used to be difficult to measure very condensed or colored sample due to multi scattering or absorption effect. Currently standard cell of ELSZ series is able to measure wide concentration range of sample. Furthermore, zeta potential of very condensed sample can be measured by FST method*
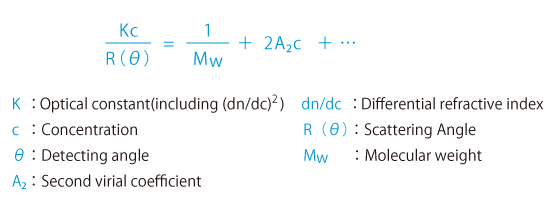
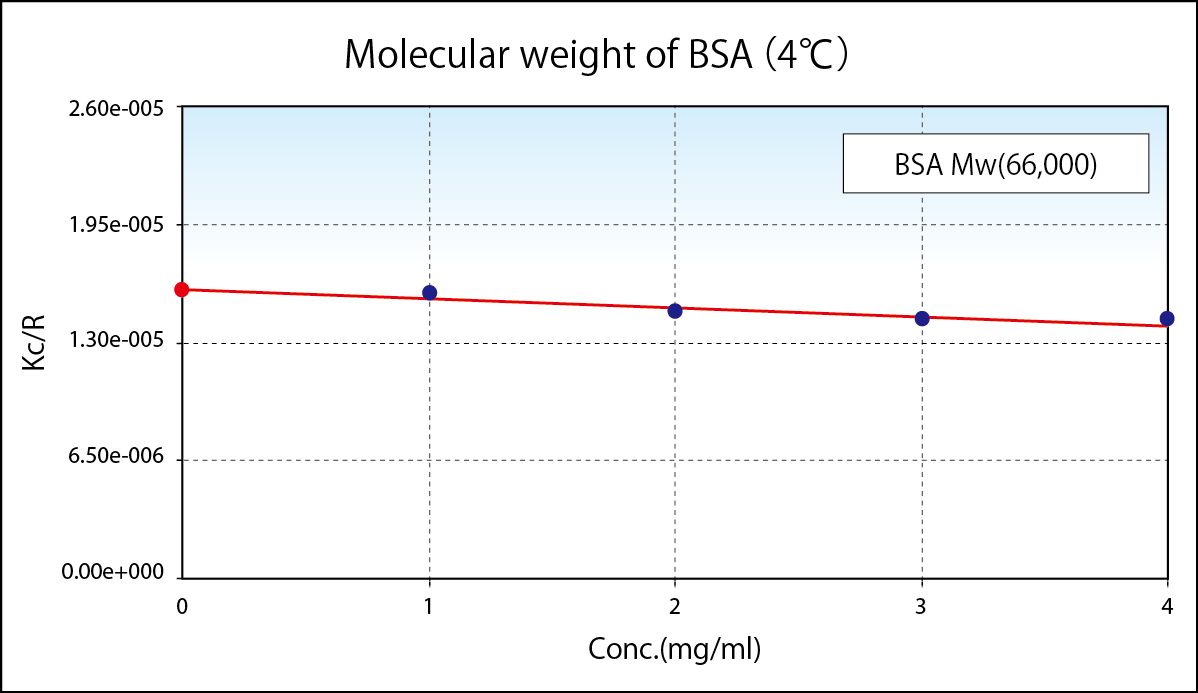
Static Light Scattering Method is renowned as the convenient method to know absolute molecular weight. As a principle, molecular weight is calculated from absolute value of scattered light obtained by irradiated light into colloidal particle. To simplify, from the bigger particle gives the stronger scattering and the smaller particle gives weaker scattering. In reality, scattering intensity to be obtained depends upon concentration, too. So the plotting concentration on horizontal and Kc/R(θ), which is equivalent as a reciprocal number of scattering intensity, on vertical after measuring scattering intensities of variable concentrations is called Debye Plot.
Since angle dependency of scattering intensity appears in the large molecular weight sample, measuring scattering intensity at variable angle(θ) provides more accuracy on molecular weight measurement and radium of gyration, which is yardstick of molecular dispersion. When measured at fixed angle, inputting expected radium of gyration, correcting as an angle dependent measurement, accuracy of molecular weight is improved
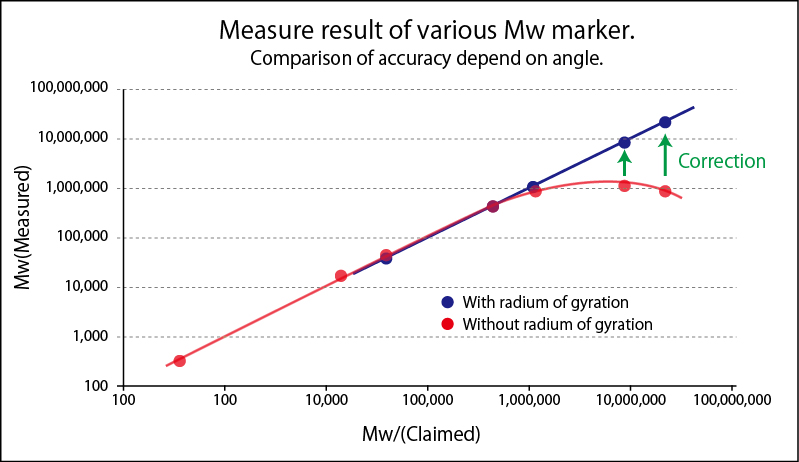
Second Virial Coefficient
Second Virial Coefficient expresses the degree of attraction and repulsion of monomers, giving the yardstick of affinity and crystallization against molecule of solvent.
- Given A2 is positive, repulsion in molecules is big in high affinity good solvent, it exists steadily.
- Given A2 is negative, affinity in molecules is big in low affinity poor solvent, it aggregates easily.
- Given A2 is zero, the solvent is called theta solvent and its temperature is called theta temperature where attraction and repulsion are proportional, causing crystallization easily.
Specification
| Principle | Particle size | Dynamic light scattering method | ||
| Zeta potential | Laser Doppler method | |||
| Molecular weight | Static light scattering method | |||
| Optics | Particle size | Homodyne system | ||
| Zeta potential | Heterodyne system | |||
| Molecular weight | Homodyne system | |||
| Light source | High power semiconductor laser | |||
| Detector | High sensitivity APD | |||
| Cells | Zeta potential: Standard cell, Micro volume disposable cell, High-conc. cell |
|||
| Particle size/Molecular weight : Rectangular cell | ||||
| Temperature | 0 ~ 90℃ (with gradient function) | |||
| Power requirements | 100V ± 10% 250VA | |||
| Size(WDH) | 380(W)×600(D)×210(H)mm | |||
| Weight | Approx. 22 kg | |||
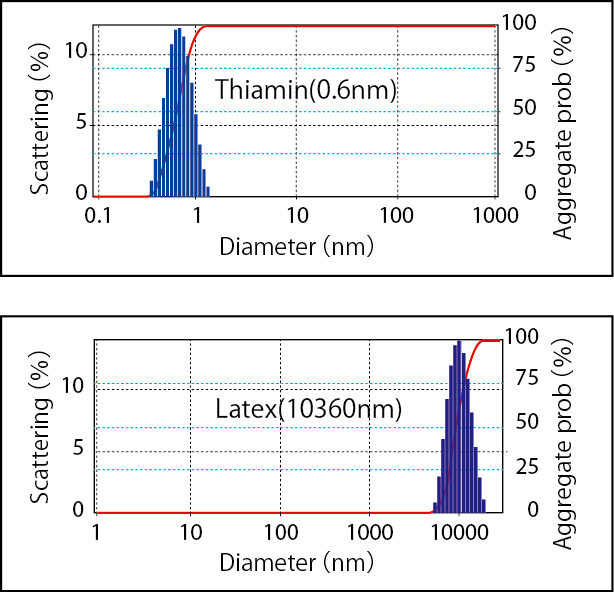
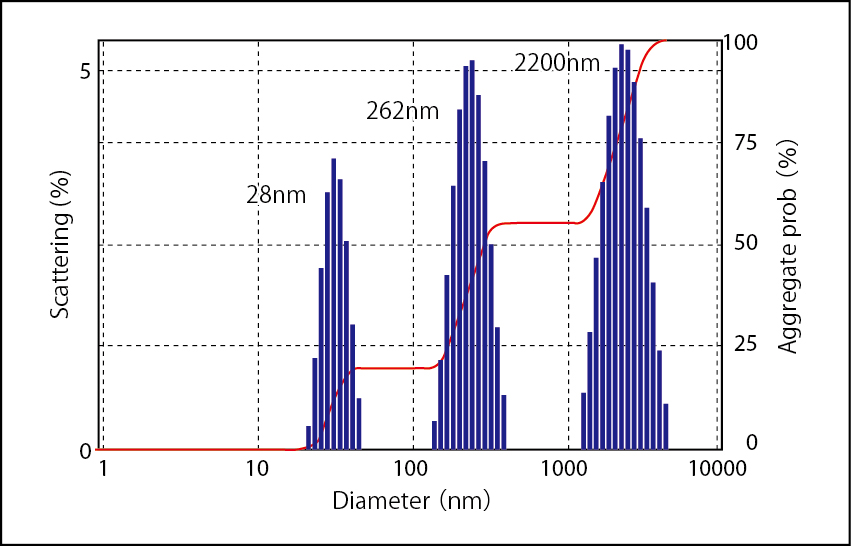
Measured Data
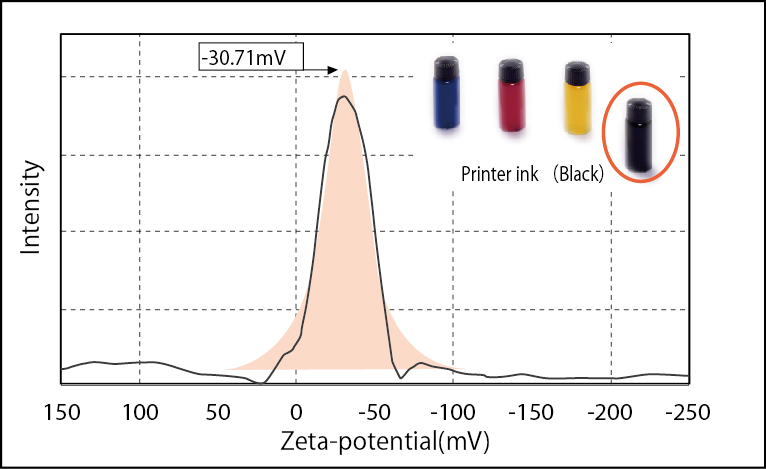
Zeta potential of ink liquid (Black)
Measure example of solid surface cell unit
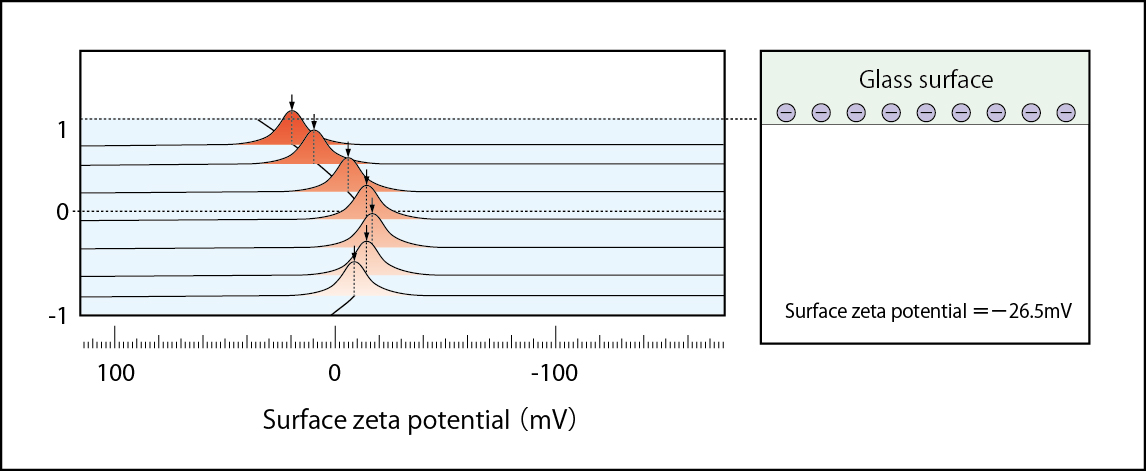
Zeta potential of negatively charged glass surface (BLANK) Zeta potential =-58.4mV:(1mM NaCl solution)
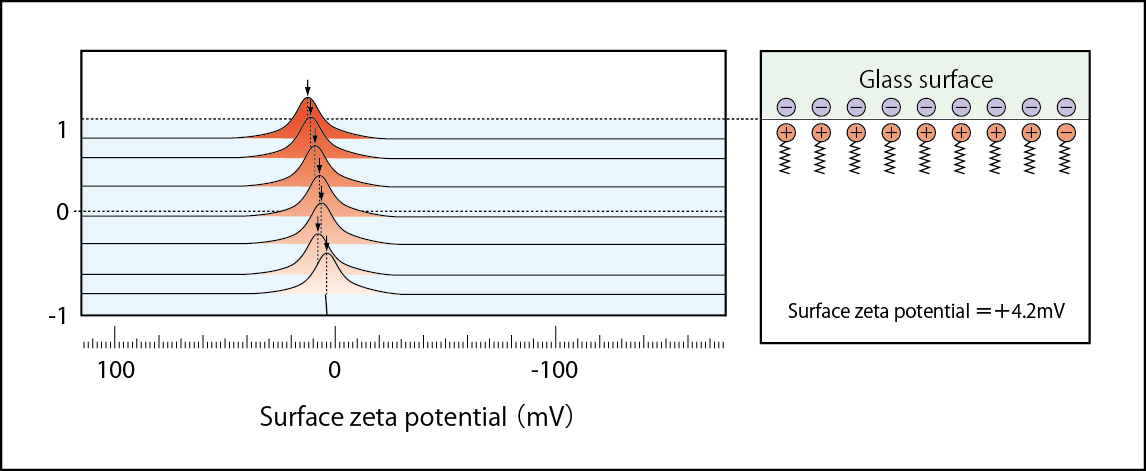
Negatively charged glass surface being neutralized by positively charged CTAB Zeta potential =+1.3mV:(1mM NaCl solution including 1×10-5mol/l CTAB)
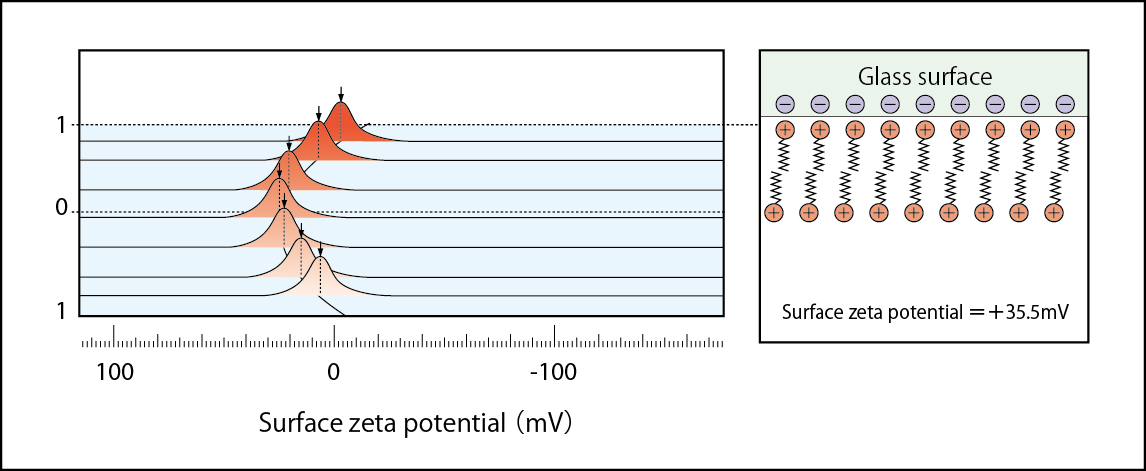
Positively charged status with the excessive CTAB adsorbed on glass surface Zeta potential =+35.5mV:(1mM NaCl solution including 1×10-4mol/l CTAB )
Measure example of micro volume disposable cell
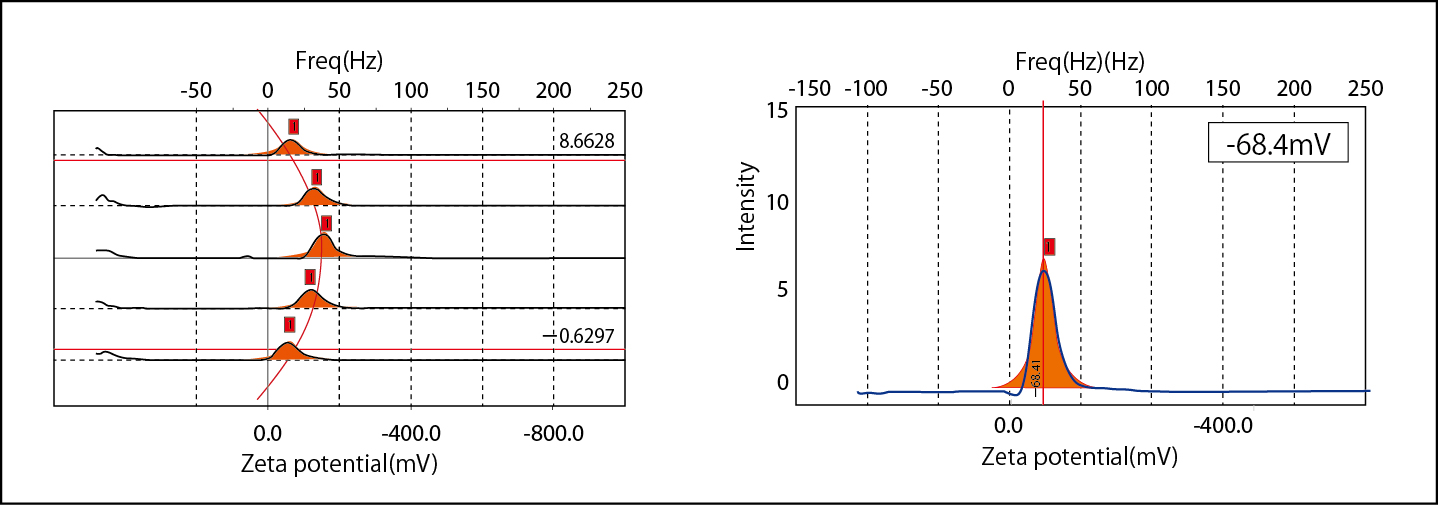
Electro-osmosis plotting and zeta potential of 100mM NaCl Latex solution
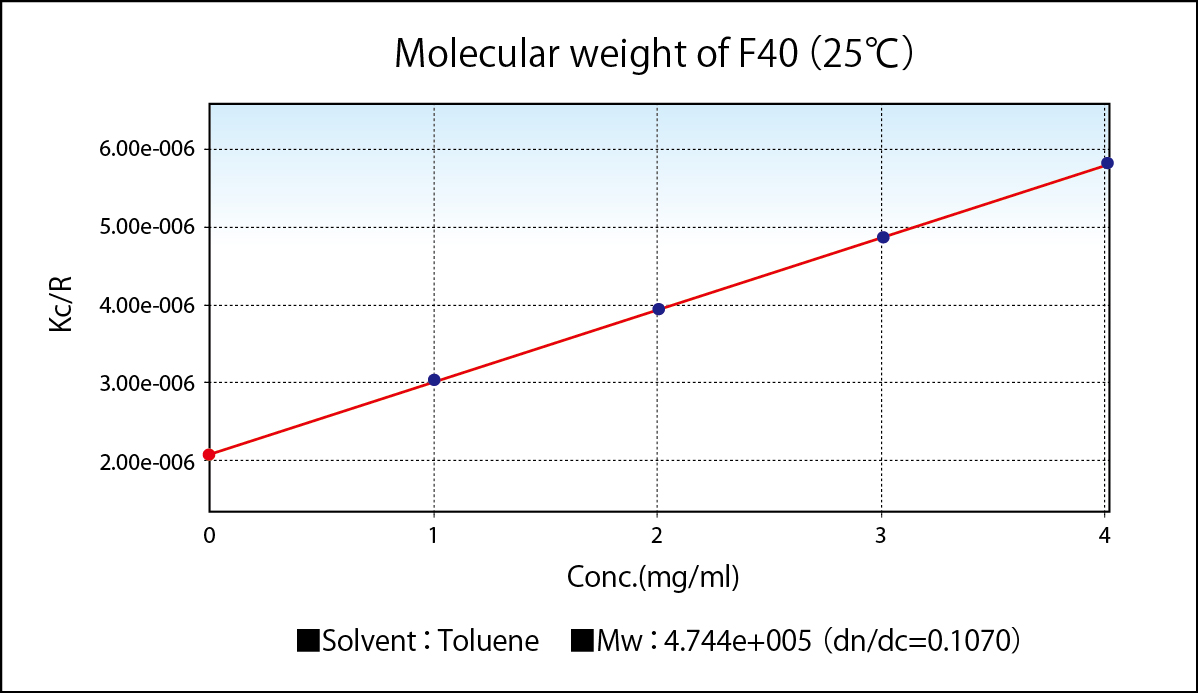
Molecular weight of F40(25℃)
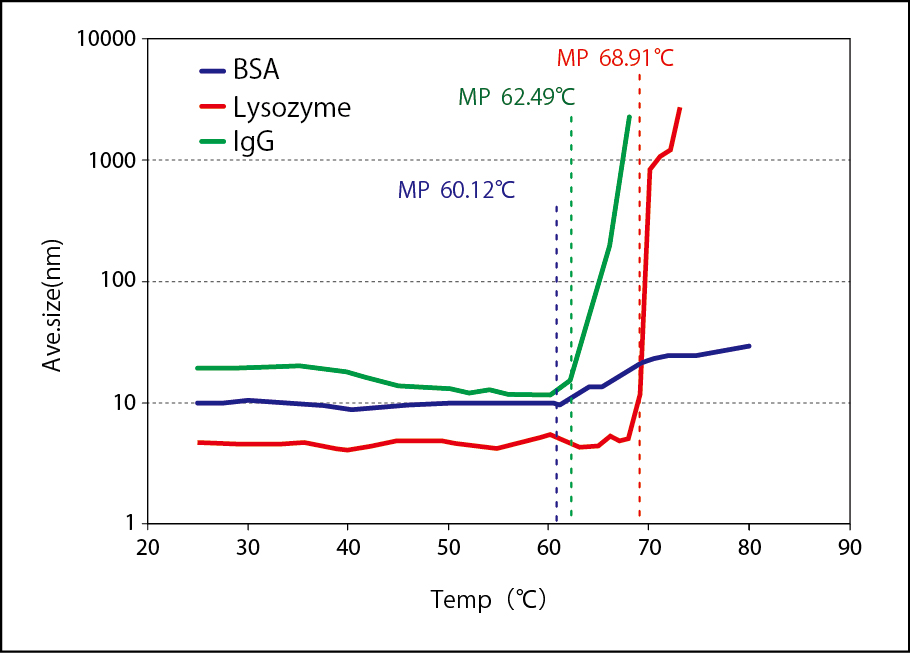
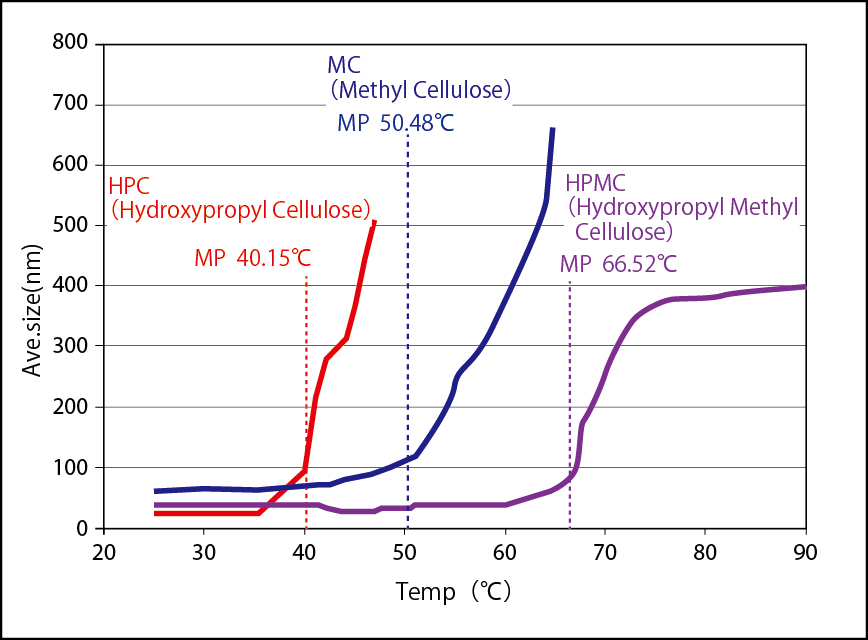
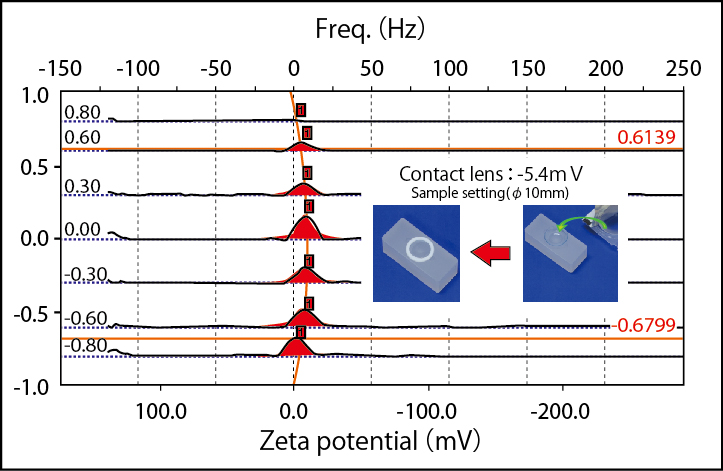
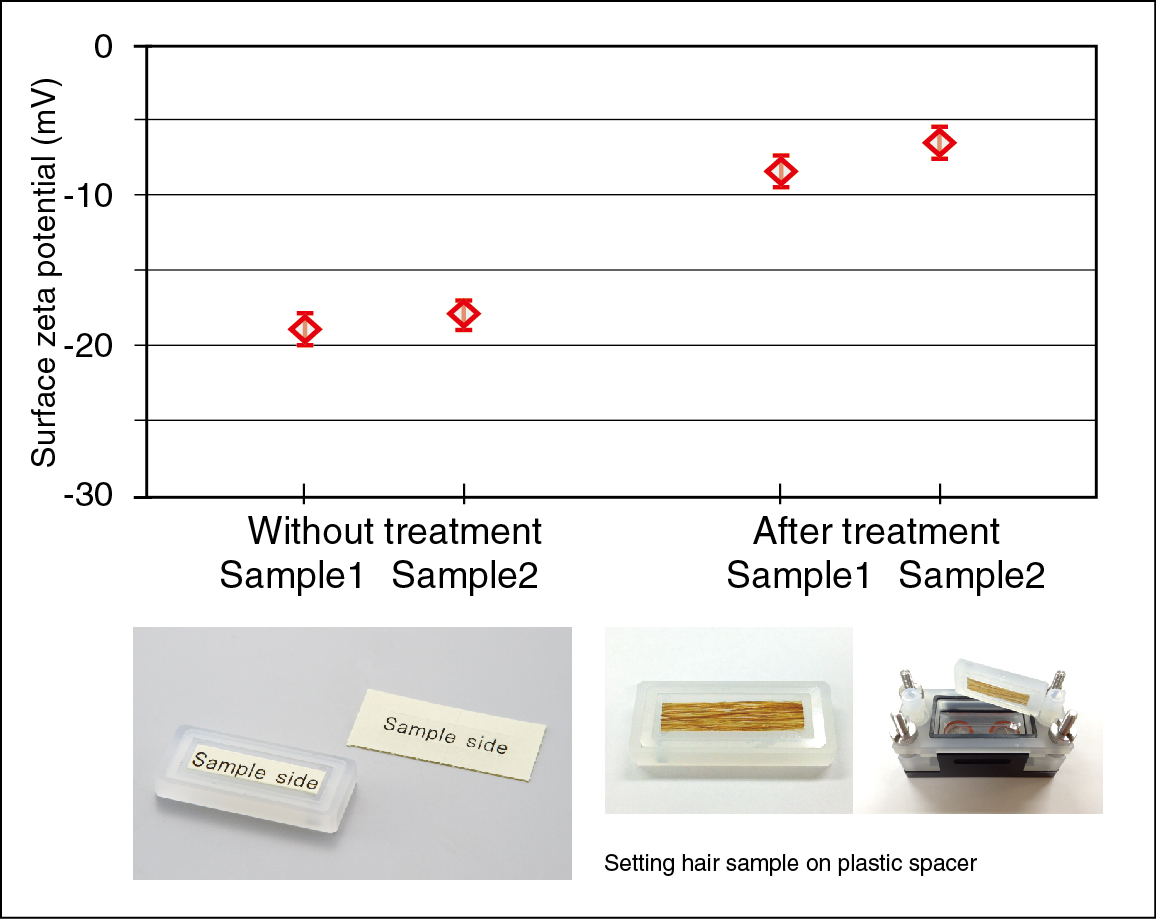
Surface zeta potential of human hair
Option
pH Titrator System(ELSZ-PT)

ELSZ + ELSZ-PT


Solid sample cell enables surface zeta potential measurement of film or plate like sample. The principle employs the electro-osmosis caused from surface charge of cell. It runs electro-osmosis assuming that one cell inside wall being replaced with solid state sample. The surface charge of solid state sample affects electro osmosis followed by analyzing with Mori-Okamoto equation to obtain zeta potential.
| Sample | Sheet or flat state sample | |||
| Sample size | Max:37×16(mm) Min:33×14(mm) Thickness:5(mm)or less |
|||
- Product
- Principle
- Specification
- Measured Data
- Option


 Close
Close





