Introduction:Zeta Potential
1.What is zeta potential?
Particles dispersed in the liquid are generally charged positively or negatively. In order to maintain electrical neutrality, ions having an opposite sign to particles gather on the surface of the particle. When a group of ions surrounds the particle surface and gathers in a spherical shape, the charged layer will be surrounded by the layer with the opposite charge. This condition is described as "an electrical double layer was formed". What electricity is stored in a condenser is considered like this.
However, the ionic layer in the liquid is not as simple as this. The distribution of ions is disturbed due to thermal motion. Therefore, the concentration of the opposite charge is high in the vicinity of the surface, and it decreases gradually as it moves away from the surface. The particles and the charged ions show the opposite distribution. In areas far enough from the particles, positive ion charge and negative ion charge cancel off to maintain electrical neutrality. For the condenser-type double layer described above, what is actually seen in the liquid is called "diffused electrical double layer". These terms describe the electrical double layer, which is the ion distribution of the opposite charged gradually blurs as it leaves the surface.
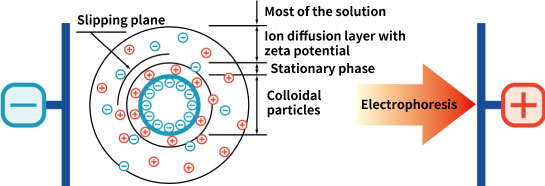
Compared to this, the ion distribution on the inner particle surface is not blurred. The diffuse electrical double layer is named with a focus on the situation of one side. This side is called the diffusion layer. The diffusion layer does not necessarily exist in the vicinity of surface. Some ions are often strongly attracted to the surface and fixed. These layers are called fixed layers. This is sometimes referred to as the Stern layer.
Mentioned at the beginning, particles dispersed in liquids often have charge. And the dispersion stability state of the particles is often determined by the charged state. In this case, what should be an indicator of the charged state of particles? Zeta potential is defined to answer this question. The problem is that it is not likely to be charged with naked particles stripped to a fixed layer, and it is unlikely to measure this directly. The charge that moves with the particles seems to directly affect the behavior of the particles, and it seems that it can be measured. It can be estimated that the particles move with the fixed layer and an inner part of the diffuse layer. The face on which this movement occurs is called a slipping plane.
Define the potential of a region that is electrically neutral enough away from the particles as zero. Zeta potential is defined as the potential of the sliding plane when measured relative to this zero point. In the case of fine particles, the higher the absolute value of the zeta potential, the stronger the repulsive force between the particles and the higher the dispersion stability of the particles. Conversely, when the zeta potential is close to zero, the particles are more likely to aggregate. Therefore, zeta potential is used as an index of dispersion stability of dispersed particles.
.jpg)
In recent years, fine particles have been often used as materials. In order to improve its functionality, research on their surface modification is actively carried out. Attempts have been made to adsorb polymer compounds, surfactants, or heterogeneous fine particles on the surface or chemically treat the surface. In addition, the conditions such as the pH value of the dispersion medium is also being examined in detail. The importance of zeta potential as an indicator of particle stability is increasing.
Fumio Kitahara, Kunio Furusawa, Masataka Ozaki, Hiroyuki Oshima,
"Zeta Potential Zeta Potential: Physical Chemistry at The Particulate Interface," Scientist, Inc., 1995.
Related Products
| Zeta-potential & Particle size Analyzer ELSZneo | |
| Zeta-potential & Particle size Analyzer ELSZ-2000 series |


 Close
Close



